
Mini-titrators for Wine Analysis
User-friendly microprocessor-based mini-titrators. Simple, fast, and affordable designed for testing total acidity levels and free and total sulphur dioxide in wine.
The Milwaukee MI455 PRO SO2 Mini-Titrator is preferred by wine makers seeking results faster with lower costs and simple operation.
The Milwaukee MI456 PRO TA Mini Titrator is preferred by wine makers seeking results faster with lower costs and simple operation.

Mi455 Mini-titrator for the determination of Free & Total Sulphur Dioxide in wine analysis
Free & Total Sulphur Dioxide
Lab Grade. Accurate. Simple. Automated.
The Milwaukee MI455 PRO Mini-Titrator makes the measurement of free and total Sulfur Dioxide simple. The MI455 PRO can be used to test for free and total SO2 in all wines, including red. Using a modified Ripper method, the MI455 PRO Mini-Titrator does not rely on color change but uses potentiometric detection. The MI455 PRO Mini-Titrator automatically dispenses the titrant, determines the endpoint, and completes the calculations in a fraction of the time needed for a manual titration. The MI455 PRO uses a preprogrammed method and specialized ORP electrode combined with an algorithm to determine the end of the titration reaction.
The Milwaukee PRO SO2 PRO Mini-Titrator offers:
Accuracy
The MI455 PRO Mini-Titrator is accurate to 5% of the reading with a resolution of 1 ppm with a range of 0-400 ppm. The titrator uses piston-driven pump for accurate dynamic dosing, a built-in magnetic stirrer for a faster reaction and lab grade platinum-ringed ORP electrode.
Simplicity
The MI455 PRO Mini-Titrator is designed to for simple one button operation with pre-standardized titrant and pre-measured reagents for easy set up.
Automated Operation
The MI455 PRO Mini-Titrator uses a precision piston style dosing pump that dynamically adjusts the dosing volume as the voltage changes. With the MI455 PRO Mini-Titrator, you get a faster result with no need for manual intervention or calculation.
Design Features
• Fast, easy to read results
• Accuracy to ±5% of reading
• Easy measurement: just measure the wine sample, add the reagents as directed, prepare ORP probe and pump, add the stabilizer and start the titration by pressing the start button
• Refillable, double junction lab grade ORP electrode for fast results with a screw on cap to ensure proper storage
• Compact design / small footprint
• Built-in stirrer is automatically maintained at a speed of 1500 rpm
• Built-in algorithm to analyze the shape of the electrode response and to determine the reaction completion that automatically completes the analysis, makes all the necessary calculations and assures a simple and effective interface for the user
• Simple and reliable peristaltic pump that ensure high dosing repeatability with the ability to calibrate with the provided Milwaukee standards
• Ability to easily measure both free and total SO2
Care & Use
ORP electrodes usually start performing poorly because of problems with the probe. The two parts of the ORP probe that cause problems are the glass sensing bulb and the reference junction.
Sensing Bulb
The glass bulb loses sensitivity with use and will eventually fail. This is true of all ORP probes.
Reference Junction
The reference junction is a small hole that allows the meter to compare the sample to a reference. The reading is generated based on the electrical difference between the sample and reference. If the junction hole gets clogged, the ORP electrode will no longer function.
You can extend the life of your ORP electrode in the following ways.
Cleaning & Storage
All ORP sensors have a limited life and response time gets longer with age. Properly maintaining your ORP electrode will give you fast, accurate results for longer.
Storage
To minimize clogging the ORP probe needs to be kept hydrated. Long periods of dry storage will damage the sensitivity of the probe. Allowing the probe to dry out may also result in the junction hole getting clogged.
Storing in ORP probe storage solution MA9015 will help address both these issues. Do not store in tap water or DI water. This will damage the sensitivity of the probe.
The best way to store the probe is with the probe's cap filled with storage solution and the cap tightened to prevent leakage. It is also recommended that to store the electrode upright to further reduce the potential for leakage.
Cleaning & Maintenance
Residue from the sample can impact the sensitivity of the sensing bulb and clog the junction. This is especially true if the sample has a lot of organic material. We recommend regularly soaking your electrode in cleaning solution MA9016. Also:
1. Inspect the electotrode for damage or scratches, if present replace the electrode.
2. Inspect the cable. The connection cable and insulation must be intact.
3. Connectors should be clean and dry.
4. Rinse off salt deposits with water or DI water.
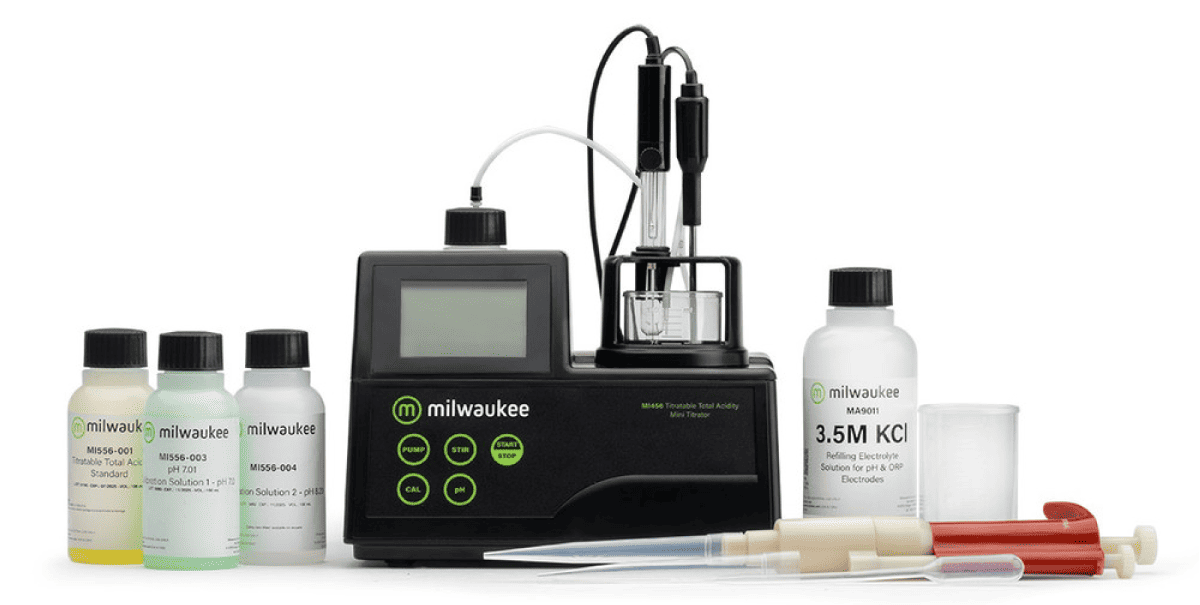
Mi456 Mini-titrator for the determination of Titratable Total Acidity for wine analysis
Titratable Total Acidity
Lab Grade. Accurate. Simple. Automated.
The Milwaukee MI456 PRO Mini-Titrator makes the measurement of Total Titratable Acidity simple. The MI456 PRO uses a neutralization reaction - the reaction between the acids found in wine and a base. This type of reaction forms the basis of titration methods of analysing acids. Titratable acidity is measured on a degassed sample at the end-point of 8.20 for Australian requirements and 7.00 to fulfil the requirements of the European Union. Both results are expressed as g/L tartaric acid. The MI456 PRO Mini-Titrator automatically dispenses the titrant, determines the endpoint, and completes the calculations in a fraction of the time needed for a manual titration. The MI456 PRO Mini-Titrator uses a preprogrammed method and lab grade pH electrode combined with an algorithm to determine the end of the titration reaction.
The Milwaukee PRO TA Mini Titrator offers:
Accuracy
The MI456 PRO Mini Titrator is accurate to 5% of the reading with a resolution of 0.1 g/L with a range of 0.0 to 25.0 g/L of tartaric acid. The titrator uses piston-driven pump for accurate dynamic dosing, a built-in magnetic stirrer for a faster reaction and lab grade pH electrode.
Simplicity
The MI456 PRO Mini-Titrator is designed to for simple one button operation with pre-standardized titrant and pre-measured reagents for easy set up.
Automated Operation
The MI456 PRO Mini-Titrator uses a precision piston style dosing pump that dynamically adjusts the dosing volume as the voltage changes. With the MI456 PRO Mini-Titrator, you get a faster result with no need for manual intervention or calculation.
Design Features
- Fast, easy to read results
- Accuracy to ±5% of reading
- Easy measurement: just measure the wine sample, add the reagents as directed, prepare pH probe and pump, add the stabilizer and start the titration by pressing the start button
- Refillable, double junction lab grade pH electrode for fast results with a screw on cap to ensure proper storage
- Automatic temperature compensation from 0.0 to 100.0 C with temperature probe included
- Compact design / small footprint
- Built-in stirrer is automatically maintained at a speed of 1500 rpm
- Built-in algorithm to analyze the shape of the electrode response and to determine the reaction completion that automatically completes the analysis, makes all the necessary calculations and assures a simple and effective interface for the user
- Simple and reliable peristaltic pump that ensure high dosing repeatability with the ability to calibrate with the provided Milwaukee standards
Care & Use
pH electrodes usually start performing poorly because of problems with the probe. The two parts of the pH probe that cause problems are the glass sensing bulb and the reference junction.
Sensing & Bulb
The glass bulb loses sensitivity with use and will eventually fail. This is true of all pH probes.
Reference Junction
The reference junction is a small hole that allows the meter to compare the sample to a reference. The reading is generated based on the electrical difference between the sample and reference. If the junction hole gets clogged, the pH electrode will no longer function.
You can extend the life of your pH electrode in the following ways.
Cleaning & Storage
All pH sensors have a limited life and response time gets longer with age. Properly maintaining your pH electrode will give you fast, accurate results for longer.
Storage
To minimize clogging the pH probe needs to be kept hydrated. Long periods of dry storage will damage the sensitivity of the probe. Allowing the probe to dry out may also result in the junction hole getting clogged.
Storing in pH probe storage solution MA9015 or pH calibration buffers will help address both these issues. Do not store in tap water or DI water. This will damage the sensitivity of the probe.
The best way to store the probe is with the probe's cap filled with storage solution or calibration buffer and the cap tightened to prevent leakage. It is also recommended that to store the electrode upright to further reduce the potential for leakage.
Cleaning & Maintenance
Residue from the sample can impact the sensitivity of the sensing bulb and clog the junction. This is especially true if the sample has a lot of organic material. We recommend regularly soaking your electrode in cleaning solution MA9016. Also:
1. Inspect the electotrode for damage or scratches, if present replace the electrode.
2. Inspect the cable. The connection cable and insulation must be intact.
3. Connectors should be clean and dry.
4. Rinse off salt deposits with water or DI water.
Calibration
The pH sensing bulbs become less sensitive over time so make sure that you are calibrating regularly to keep your MI456 Mini-Titrator readings accurate. Also, calibrate after cleaning or a long period of storage.
Need more information / Questions?
Need more information / Questions?
Please contact us by phone or email:
Freephone 0800 724 633
Or use the contact form below:
A to Z Product Listing
A to Z Product Listing
Copyright © 2026, Scienze. All rights reserved.


